- Blog
- How to create multiple ids for fluenz online
- Ark survival evolved key free
- Least expensive macbook
- Facebook template for microsoft word 2003
- Microsoft xbox 360 driver download windows 7
- Free web buttons book an event
- What does org stand for victoria police
- Itunes mac sleep timer
- 2006 macbook hard drive replacement
- Mayflash gamecube controller adapter for pc
- Kumari 21 f movie online hd free
- Free plant vs zombie download
- How to install excel solver add in
- Macromedia dreamweaver 8 tutorial
- What is abgx360
- Full size modern periodic table pdf
- Download slingbox app for chorme
- What is the latest apple mac update do
- What does darthmod for empire total war
- Microsoft server management tools windows 10
- Selena gomez revival album full
- Kohler campbell piano serial number 224511
- Procreate for pc windows 10
- Wireless adapter for mac laptop
- Premium ccgen
- Photoshop cs6 mac sierra

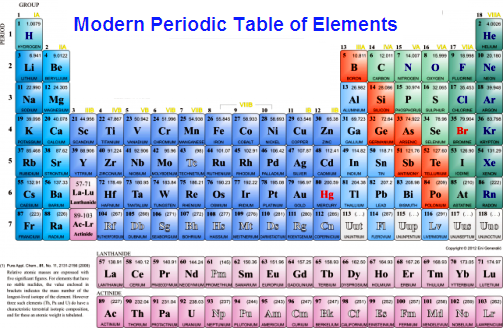
Describe and model the structure of the atom in terms of the nucleus, protons, neutrons and electrons comparing mass and charge of protons neutrond and electrons. Use the Periodic Table to predict the ratio of atoms in compounds of two elements. Unit 1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.Unit C1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.(g) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups.2.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.(h) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups.1.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.(a) elements being arranged according to atomic number in the Periodic Table.
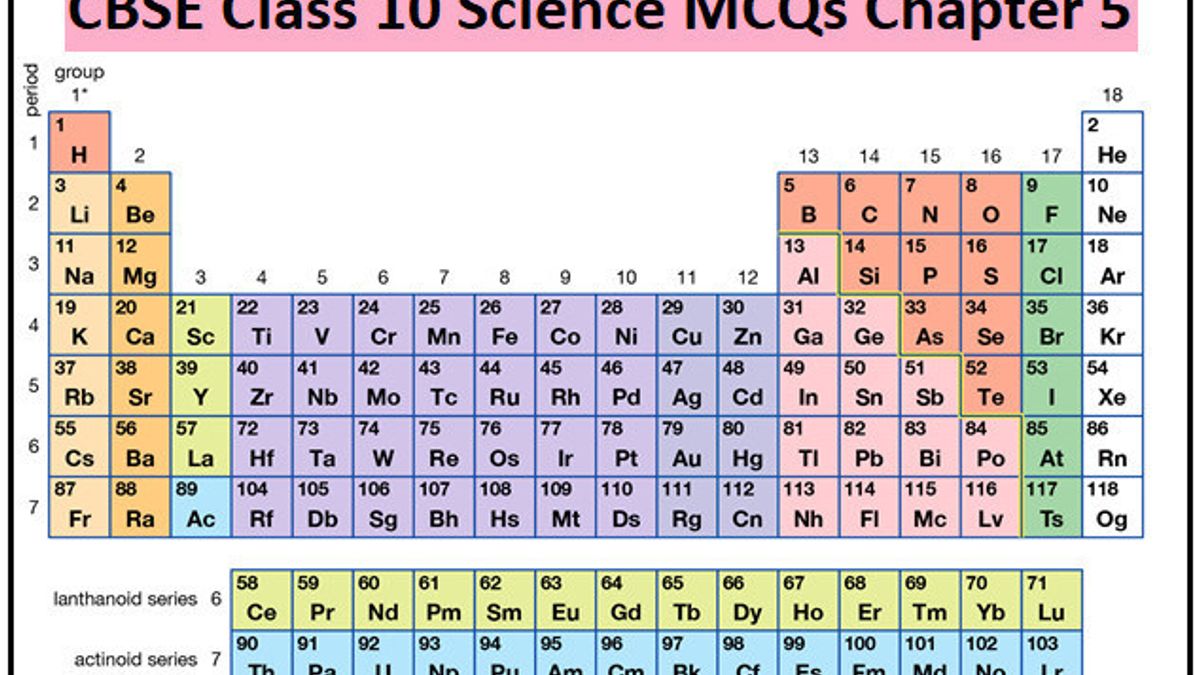
Unit 1: THE LANGUAGE OF CHEMISTRY, STRUCTURE OF MATTER AND SIMPLE REACTIONS.The Periodic Table can be used to determine whether an element is a metal or non-metal.Atomic structure and bonding related to properties of materials.Elements are arranged in the periodic table in order of increasing atomic number.Unlike theories such a specific properties. Hamied Inspirational Chemistry Programme The isotopes of same element will be given different focus if atomic mass is the basis of classification.Introductory maths for higher education.The physics of restoration and conservation.
- Blog
- How to create multiple ids for fluenz online
- Ark survival evolved key free
- Least expensive macbook
- Facebook template for microsoft word 2003
- Microsoft xbox 360 driver download windows 7
- Free web buttons book an event
- What does org stand for victoria police
- Itunes mac sleep timer
- 2006 macbook hard drive replacement
- Mayflash gamecube controller adapter for pc
- Kumari 21 f movie online hd free
- Free plant vs zombie download
- How to install excel solver add in
- Macromedia dreamweaver 8 tutorial
- What is abgx360
- Full size modern periodic table pdf
- Download slingbox app for chorme
- What is the latest apple mac update do
- What does darthmod for empire total war
- Microsoft server management tools windows 10
- Selena gomez revival album full
- Kohler campbell piano serial number 224511
- Procreate for pc windows 10
- Wireless adapter for mac laptop
- Premium ccgen
- Photoshop cs6 mac sierra
